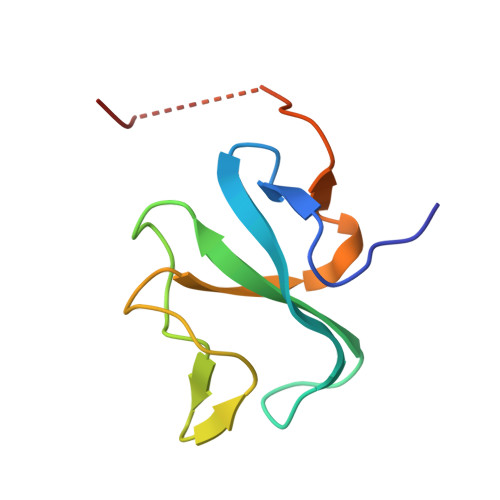
Structural basis for tubulin recognition by cytoplasmic linker protein 170 and its autoinhibition | PNAS

The microtubule plus-end-tracking protein CLIP-170 associates with the spermatid manchette and is essential for spermatogenesis

Ninein is essential for apico-basal microtubule formation and CLIP-170 facilitates its redeployment to non-centrosomal microtubule organizing centres | Open Biology
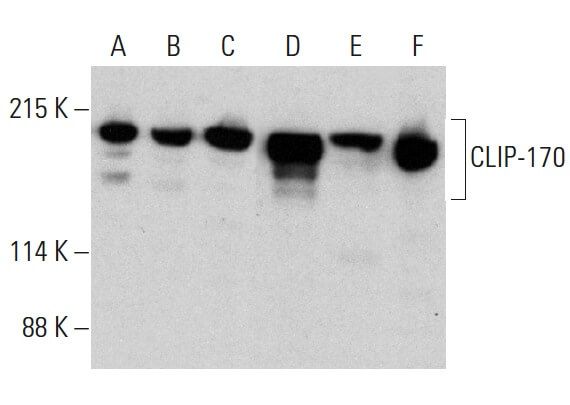
CLIP-170S is a microtubule +TIP variant that confers resistance to taxanes by impairing drug-target engageme

AMPK controls the speed of microtubule polymerization and directional cell migration through CLIP-170 phosphorylation | Nature Cell Biology
Overexpression of the microtubule-binding protein CLIP-170 induces a +TIP network superstructure consistent with a biomolecular condensate | PLOS ONE

Structural basis for tubulin recognition by cytoplasmic linker protein 170 and its autoinhibition | PNAS

The CLIP-170 N-terminal domain binds directly to both F-actin and microtubules in a mutually exclusive manner - Journal of Biological Chemistry
CLIP-170S is a microtubule +TIP variant that confers resistance to taxanes by impairing drug-target engageme
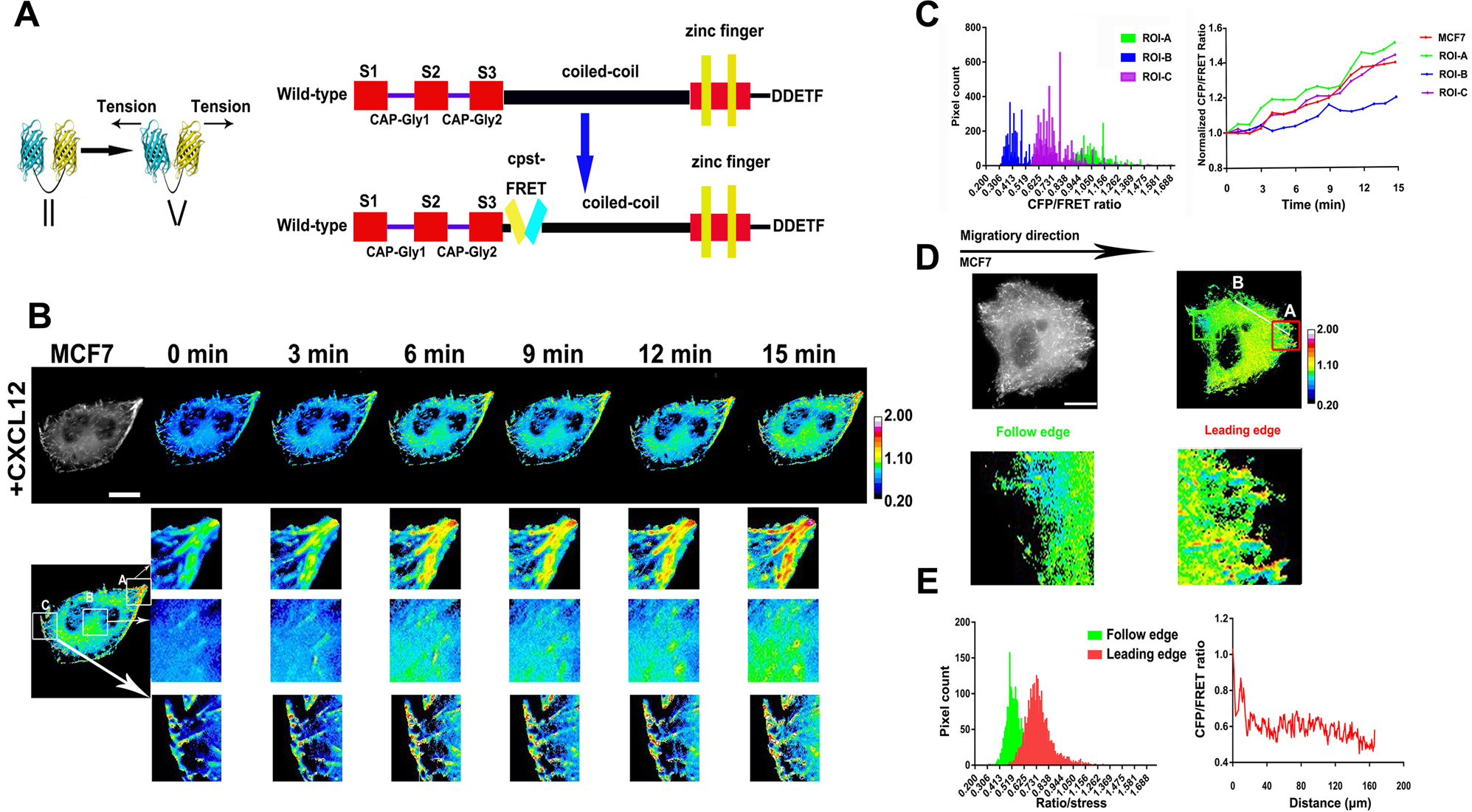
Tension of plus-end tracking protein Clip170 confers directionality and aggressiveness during breast cancer migration | Cell Death & Disease
Overexpression of the microtubule-binding protein CLIP-170 induces a +TIP network superstructure consistent with a biomolecular condensate | PLOS ONE

HIV‐1 capsids mimic a microtubule regulator to coordinate early stages of infection | The EMBO Journal

The microtubule plus-end-tracking protein CLIP-170 associates with the spermatid manchette and is essential for spermatogenesis
Potential mechanisms of microtubule plus-end tracking. (A) Motor-driven... | Download Scientific Diagram

α-Tubulin Tyrosination and CLIP-170 Phosphorylation Regulate the Initiation of Dynein-Driven Transport in Neurons - ScienceDirect
Overexpression of the microtubule-binding protein CLIP-170 induces a +TIP network superstructure consistent with a biomolecular condensate | PLOS ONE
Overexpression of the microtubule-binding protein CLIP-170 induces a +TIP network superstructure consistent with a biomolecular condensate | PLOS ONE